MBBR Media
Transcend CleantecPvt Ltd is the supplier of specialized and patented high-performance MBBR media, bio media,/fab media/ reactor media called TRANSCHIP. Transcend Cleantecpvt Ltd has an exclusive agreement with MUTAG GERMANY AND is a well recognized name as a supplier and distributor of quality MBBR media, with its business operations running from Pune, India. TRANSCHIP is available in two diff types : Virgin PE and recycled PE alternative. Removal rates of Transchip are upto 13 times higher than with other “conventional” carriers. The Transchip is a high performance MBBR carrier and provides a protected active surface area of aprrox 5,500 m2/m3 for the immobilization of micro organisms which are in charge of the different biological water treatment processes. Its application is characterized by extremely high removal rates and reliable process stability. The pores of the TRANSCHIP are permanently filled with active biomass since substrate and/or oxygen can diffuse into the biofilms down to a depth of approx. 0.5 mm on both sides of chips.
The MBBR media system is appraising a biofilm process. Other conventional biofilm processes for wastewater treatment is also known as trickling filter, rotating biological contactor (RBC) and biological aerated filter (BAF). TRANSCHIP MUTAG MEDIAprocedure in commonneeds less space than activated sludge structures because the biomass is more focused, and the efficiency of the system is less dependent on the final sludge partition. A disadvantage with other biofilm processes is that they experience bio clogging and a build-up Microbial loss.
Enquiry NowProduct Specification:
| Surface Area | 5500 m2/m3 |
|---|---|
| Height | 1.1 mm |
| Diameter | 30 mm |
TRANSCHIP is used in the following biological treatment processes:
- Denitrification
- Nitrification
- COD removal
- ANAMOX
The essential benefits at a glance
- up to 5,500 m²/m³ of surface area or 15% more perfomance
- outerdiameter: approx. 30 mm
- blockage-free, large-pored retention screens (bore diameter: 20 mm)
- optimized motion characteristics in the moving bed reactor
- diffusion of substrate and O2 into the complete biomass
- stable outer ring for protection from mechanical impacts
- THE carrier media of the future for most compact and efficient biological wastewater treatment systems, or for optimization of the biological removal performance.in comparison more favorable
- Particularly suitable for municipal/domestic sewage and for solid-rich waste waters.
- Higher effective sludge retention time (SRT) which is favorable for nitrification
- Responds to load fluctuations without operator intervention
- Lower sludge production
- Less area required

- Today, “Transcend Cleantec” has developed a well-established presence in the market because of its quality products and strong network of distributor chain. Further, its transparent and ethical business transactions as well as well-developed logistics network also allows it to promptly serve clients as per the placed orders.Transchip already has installations in wide range of effleunts such as, Sun Pharmaceuticals Ltd – AhmedanagrTATA BSL LTD.Bhushan Power LtdNTPC Ltd- Varanasi Leachate treatment plantVenkateshwara Hospitals Ltd,Delhi (Replaced Japanese Media with our media) Iros Hotels Ltd. Delhi
- Mutag BioChip 30™, the ultimate MBBR carrier for biological wastewater treatmentDr.-Ing. Markus Geiger, Bernd Rauch (Multi Umwelttechnologie AG)German company Multi Umwelttechnologie AG has over 18 years of experience inthe operation of carrier media used for immobilising microorganisms in biological wastewater treatment plants and systems. During this time, we have used almost every known biofilm carrier in numerous different large-scale plants, in different types of wastewater as well asin different fields of application. The knowledge that we gained in result, along with awide data base, enables us to make a well-founded assessment of the specific performance data of several biofilm carrierswhich were in some cases extremely different from each other. As Multi Umwelttechnologie AG has at no time been dependent from any particular supplier, we were able to implement targeted optimization either of the carrier itself or of the process conditions, based on the operational experience we gained. Here, the emphasis was on minimising operational problems arising from the shortcomings of “conventional” biofilm carrier media,whichare not to be neglected nowadays. The thorough implementation of our requirements on an optimal biofilm carrier led to one final result named Mutag BioChip™. From our point of view, it is currently the “best carrieravailable” notably for purifying types of wastewater that are difficult to treat.
1.How the Mutag BioChip 30™ works
The particularfunctionality as well as the efficiency of the Mutag BioChip 30™ canbe explained bestby orientating on the characteristic MBBR process conditions (MBBR = moving bed biofilm reactor). A crucial prerequisite for the biological removal of wastewater components in the MBBRprocess is the immobilisation of the microorganisms on the surface of carrier elements. Here,the efficiency of the biological removal is determined by the carrier’s “active” surface area. This comes along with the following carrier requirements: Firstly, there must be available sufficient protected effective surface area in order to enable the microorganisms to survive and to reproduce themselves in these areas; and secondly, it is necessary to realise a maximum of mass transfer (substrate, oxygen, metabolic products) between microorganisms and wastewater. At first glance,the simultaneous fulfilling of both of these requirements seems to be a procedural contradiction, but this can be disproved as follows:
The error of maximising the volumetric surface area (in m²/m³ of carrier elements
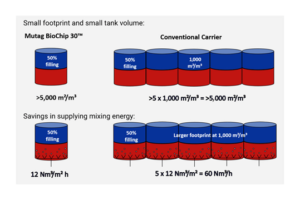
- Firstly, the error of maximising the volumetric surface area (in m²/m³ of carrier elements).Of course, it is possible to produce carrier media possessing an extremely high porosity, but it is also a fact that these pores must be accessible to the microorganisms as a potential habitat. It is easy to understand that this is hardly possible for cavities inside a carrier. If, as it is commonly the case, the transformation efficiency is correlated to the porosity, this represents a fatal distortion of the actual relations.
Limitation of the biodegradation by “thick” biofilms due to clogging and “non-biological” impurities.
2.Operational results
Of course, any theoretical considerations require the verification of the projectedbenefits within a confidence-building time frame. So far, we are able to fall back on eightyears worth of operational experience with the Mutag BioChip 30™ and its predecessor Mutag BioChip ™; during thistime it was possible in some cases to operate systems in parallel in order to compare the Mutag BioChip™with “conventional” carrier media. Unfortunately, not all operational results can be listed at this point, as this would understandably exceed the scope of this article. However, on the basis of the selected case examples, it is possible to meaningfully document the efficiency of the Mutag BioChip™. We consider it important to point out that the results listed herein are of fundamental nature, allowing transferring them to other types of wastewater and treatment applications.
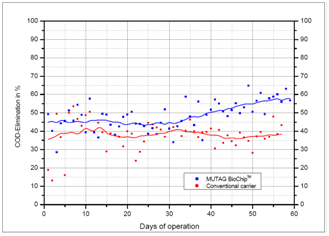
- Related to the direct comparison of biodegradation efficiency, the upgrading of a high-load stage provided us with valuable results inthe treatmentof the effluent of a paper mill. In this case, the major task was to increase the system capacity from 25,000 kg COD/d up to 50,000 kg COD/d, and to keep the paper production running throughout the complete civil works phase. For this reason, the following procedure was chosen: Initially, the existing high-load reactor filled with conventional carriers was kept in operation, whereas a similar second tank waserected in parallel. After the installation of the mechanical equipment (Figures 6 and 7), around 7% of the old reactor’s carrier volume in the form of Mutag BioChip™ carrierswere put into the new high-load reactor in order to get an initial orientation;subsequently, both high-load stages were fed with the similar wastewater volume flow rate.
- Based on the positive findings gained here, the BioChip volume was increased in a second step to 11% related to the “conventional”carrier volume that would normally have been required. Figure no. 8 shows the results that were obtained afterwards. It is clearly obvious that the Mutag BioChip™ reactor now attained the same removal efficiency as the old reactor did, so there was no need to further increase the BioChip volume. In direct comparison with the conventional carrier media, the increase in the degradation efficiency by the factor of nearly 10 achieved by Mutag-BioChip™ was proven clearly and impressively. Based on both these findings and the stable operating results of the new Mutag BioChip™ high-load stage, the old reactor was put out of operation and modified.
- Now, both stages are operated with the Mutag BioChip™ technology and reliably attain the required treated effluent parameters. With regard to the scope of the entire upgrading, it is worth mentioning for the sake of completeness that at least 1,000 m³/h of sewage water will be treated herein the future whereas anupgrade of the low-load activated sludge stage is alsoenvisaged. As a second example and as a further proof of the efficiency of the Mutag BioChip™ also in nitrogen elimination, its use in the nitrification stage ofa coke oven effluenttreatment plantwill be described in the following. Coke oven effluents are not only regarded as difficult to treat –acc. to our experience they can be biologically purifiedby means of multiple biological stage systems only.
- Here, particular biodegradation rate requirements have to be fulfilled; this applies especiallywhen taking into consideration the reactor sizes that need to remain within implementable as well as controllable dimensions. Suchlike plants using Mutag BioChip™ carriers have been in continuous operation for more than four years. Converted into population equivalents, the largest of them have a nitrification capacity of around 100,000 PE and 55,000 PE. The BioChip’s superiority can be observed here, too. Removal rates of 4-5 kg NH4-N per m³ of carrier volume are reliably attained at a constant level, even though the Mutag reactors are smaller by a factor of 5 than the activated sludge tanks which are usually required.
MBBR media for Recirculating Aquaculture system in fisheries
- Best performance in ammonia and nitrite removal- scientifically tested!
- Old dead bacteria are constantly replaced by young heavy fedding bacteria
- Protective Surface Area of 5500m2 -highest among all biocarriers!
- Small footprint - greatly reduce space required
- Less maintanance due to self-cleaning by shear force,never clogging!
- High Durability- High quality material that prevents abrasion!
- Extremely low energy consumption
- Increase oxygen content in the water
- 100% safe to use,BPA-free,No carcinogens leaching