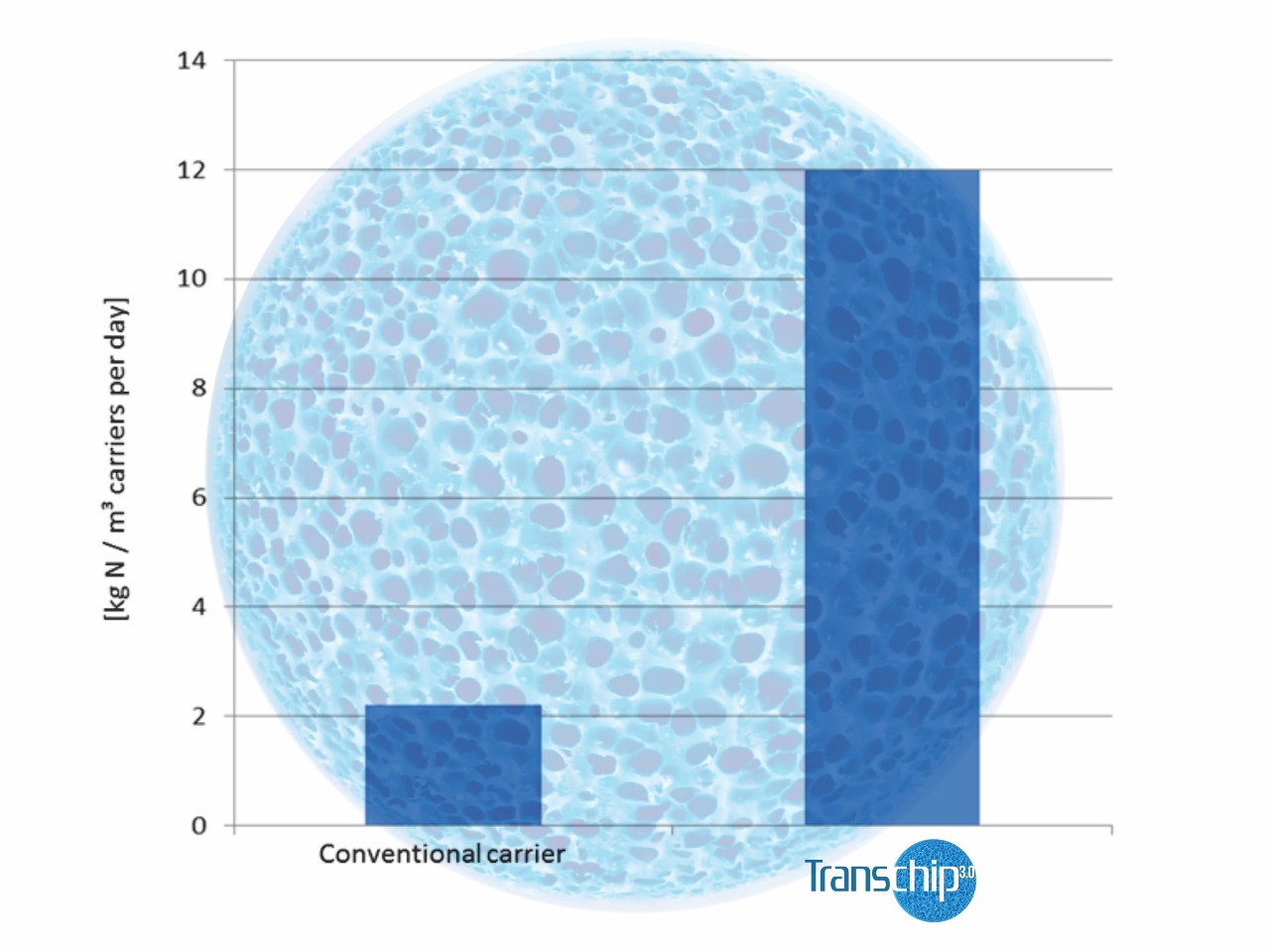
Removal rates of Transchip 3.0 are up to 13 times higher than with “conventional” carriers.
Virgin PE(Blue or White color)
Transchip 3.0
Recylced PE Alternate
Echochip 3.0

Diffusion depth from each side approx. 0.5 mm

The Transchip 3.0 MBBR Media is a high-performance MBBR carrier and provides a protected active surface area of ~ 5,500 m²/m³ for the immobilization of micro-organisms which are in charge of the different biological water treatment processes. Its application is characterized by extremely high removal rates and reliable process stability.
Cross section Transchip 3.0 MBBR Media

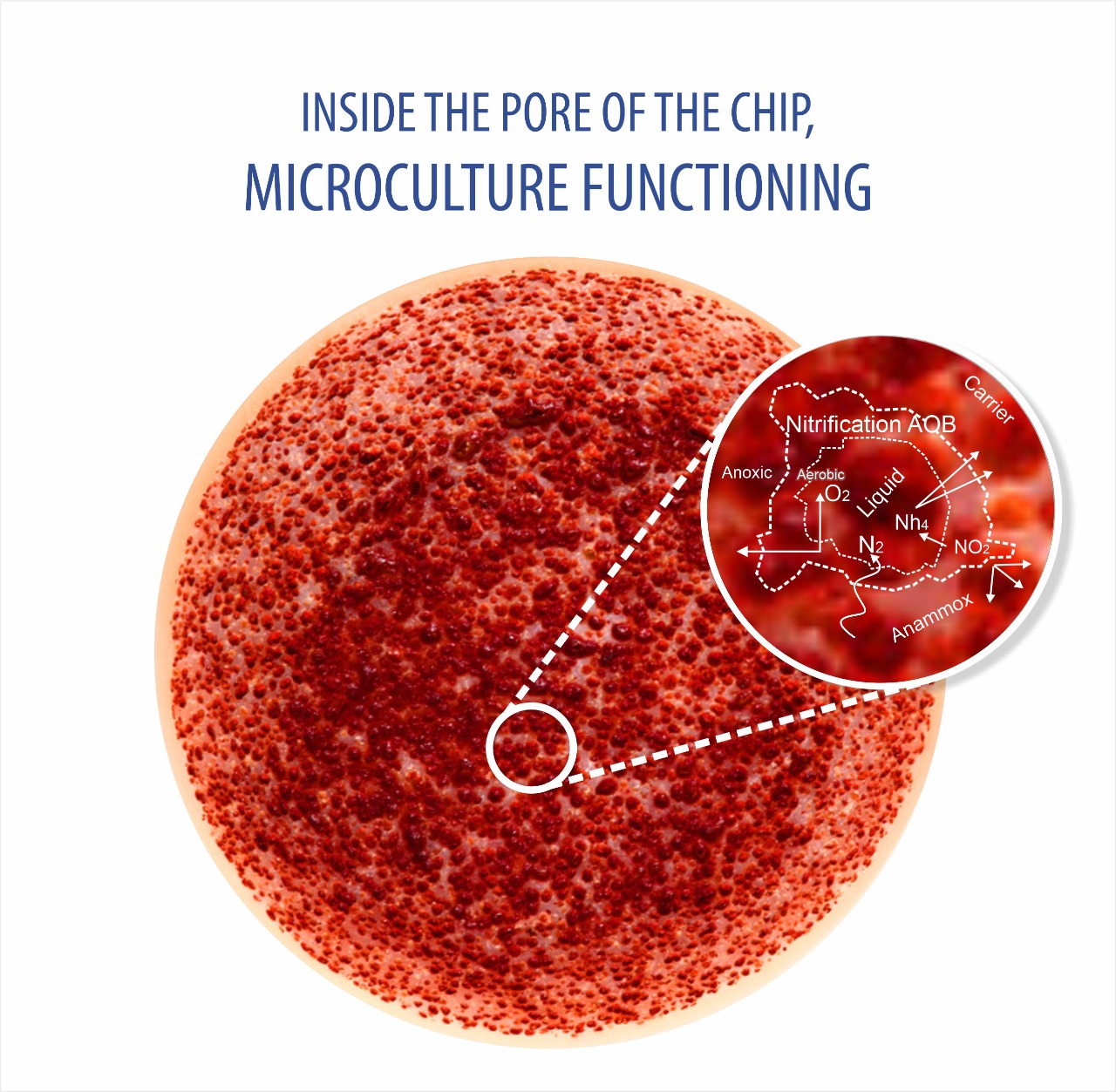
The pores of the Transchip 3.0 MBBR Media are permanently filled with active biomass since substrate and/or oxygen can diffuse into the biofilms down to a depth of approx. 0.5 mm on both sides of the chips.
~ 5,500 m²/m³

THE carrier media of the future for most compact and efficient biological water treatment system
- best possible removal performance
- remarkably stable processes
- small footprints
- ideal for easy retrofit & upgrade
- high grade quality
- long life material, no wear
- best price-performance ratio
3D-engineering by Transchip 3.0 MBBR Media

Based on more than 20 years of experience, Transcend Cleantec offers beyond conventional technology for biological treatment
3D-engineering by Transchip 3.0 MBBR Media
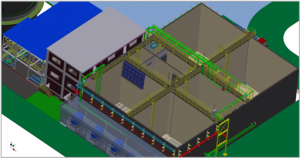
The TRANSCHIP 3.0 MBBR Technology™ is a proven biological treatment process with numerous WWTP operators world wide. A lot of plants using the TRANSCHIP 3.0 MBBR Technology™ have been designed and engineered in basic and detail by TRANSCHIP’S high-qualified engineering team.
TRANSCHIP 3.0 MBBR Technology™

- pulp & paper industry
- municipal plants
- coke & steel production
- petro and oil industry
- food and beverage industry
- pharmaceutical & chemical industry
- aquaculture industry and fish-farming
Transchip 3.0 MBBR Media with planctomycetes

TRANSCHIP 3.0 is used in the following biological treatment processes:
- Denitrification
- Nitrification
- COD removal
- ANAMMOX
Transchip 3.0 MBBR Media with biofilm

Aeration system

Carrier retention screen

Advantages of Transchip 3.0 MBBR Media
- protected active surface: ~ 5,500 m²/m³
- high quality PE material (virgin)
- optimal pore system and –structure
- less carrier volume needed
- small footprint system
- simple operation
- thin, controlled biofilms
- optimal diffusion of O2 and substrate
- easy upgrading or retrofitting of existing plants
- large diameter (30 mm; 1“)
- no abrasion & wear
- proven technology & know-how
- “Made in Germany“, developed and sold by Transcend Cleantec Pvt. Ltd. in india.
- process support for calculations
- extremely high removal capacity due to maximized concentration of active biomass in smallest possible reaction volume
- perfectly suitable for the operation with blocking-resistant and large-perforated retention screens (perforation dia. 20mm)
~ 5,500 m²/m³

THE carrier media of the future for most compact and efficient biological water treatment system
- best possible removal performance
- remarkably stable processes
- small footprints
- ideal for easy retrofit & upgrade
- high grade quality
- long life material, no wear
- best price-performance ratio
How the Transchip 3.0 MBBR Media Works
The particular functionality as well as the efficiency of the Transchip 3.0 can be explained best by orientating on the characteristic MBBR process conditions (MBBR = moving bed biofilm reactor). A crucial prerequisite for the biological removal from wastewater components in the MBBR process is the immobilisation of the microorganisms on the surface of carrier elements. Here,the efficiency of the biological removal is determined by the carrier’s “active” surface area. This comes along with the following carrier requirements: Firstly, there must be available sufficient protected effective surface area in order to enable the microorganisms to survive and to reproduce themselves in these areas; and secondly, it is necessary to realise a maximum of mass transfer (substrate, oxygen, metabolic products) between microorganisms and wastewater. At first glance,the simultaneous fulfilling of both of these requirements seems to be a procedural contradiction, but this can be disproved as follows:
The error of maximising the volumetric surface area (in m²/m³)of carrier elements
- Firstly, the error of maximising the volumetric surface area (in m²/m³ of carrier elements).Of course, it is possible to produce carrier media possessing an extremely high porosity, but it is also a fact that these pores must be accessible to the microorganisms as a potential habitat. It is easy to understand that this is hardly possible for cavities inside a carrier. If, as it is commonly the case, the transformation efficiency is correlated to the porosity, this represents a fatal distortion of the actual relations.
- This is different in the case of the Transchip 3.0 Here, a relatively thin and mainly open carrier provides an extremely large surface area in which the microorganisms can form colonies in protected pores, whereas they still remain in intensive contact with the surrounding fluid (wastewater). Consequently, the microorganisms can be optimally supplied with nutrients and the metabolic products are efficiently removed from them;this explains at least partially the effectiveness and high biodegradation efficiency of the Transchip 3.0 . Expressed in figures: the active surface of the Transchip 3.0 is up to5,500 m²/m³(1,677 ft²/ft³)
- Even in cases where a high microbial population density can be established on a carrier; if the structure and / or geometry of the carrier should beinappropriate, the mass transfer into the “deeper” layers of the biofilm is being reduced. Consequently, the biodegradation efficiency of the immobilised biological system is continually reduced during the operation. Figures show how this can look likewith various carrier types and carrier geometries. The consequences resulting from clogged carriers are easily conceivable. However, the countermeasure is very simple. The special geometry of the Transchip 3.0 enables the hydraulic shear forces acting on the outer surface to intensify, and a self-cleaning process is initiated which constantly renews the carrier’s active surface. This process effectively prevents from the limitation of the biodegradation efficiency which might be caused by mass transfer obstacles.
- Problems with the distribution and mixing-in of the carrier are prevented by the parabolic shape. In order to optimise the mass transfer (as already mentionedbefore), the Transchip 3.0 is shaped like a parabolic disc. Although it has hence an uncontrollable motion profile (at least seen from a flow-mechanical point of view), the latter has proven to turn out to be extremely positive in this case. In addition to the increased level of turbulence which directly leads to an increase in the mass transfer, the mobility of the individual carrier elements is effectively increased in the cluster. This results in a homogeneous distribution of the carriers within the entire reaction tank volume whereas the formation of any “dead zones” is effectively reduced.
Limitation of the biodegradation by “thick” biofilms due to clogging and “non-biological” impurities.
Operational results
Of course, any theoretical considerations require the verification of the projected benefits within a confidence-building time frame. So far, we are able to fall back on eight years worth of operational experience with the Transchip 3.0 and its predecessor during this time it was possible in some cases to operate systems in parallel in order to compare the Transchip 3.0 with “conventional” carrier media. Unfortunately, not all operational results can be listed at this point, as this would understandably exceed the scope of this article. However, on the basis of the selected case examples, it is possible to meaningfully document the efficiency of the Transchip 3.0. We consider it important to point out that the results listed herein are of fundamental nature, allowing transferring them to other types of wastewater and treatment applications.
- Related to the direct comparison of biodegradation efficiency, the upgrading of a high-load stage provided us with valuable results inthe treatmentof the effluent of a paper mill. In this case, the major task was to increase the system capacity from 25,000 kg COD/d up to 50,000 kg COD/d, and to keep the paper production running throughout the complete civil works phase. For this reason, the following procedure was chosen: Initially, the existing high-load reactor filled with conventional carriers was kept in operation, whereas a similar second tank waserected in parallel. After the installation of the mechanical equipment (Figures 6 and 7), around 7% of the old reactor’s carrier volume in the form of Transchip 3.0 carrierswere put into the new high-load reactor in order to get an initial orientation;subsequently, both high-load stages were fed with the similar wastewater volume flow rate.
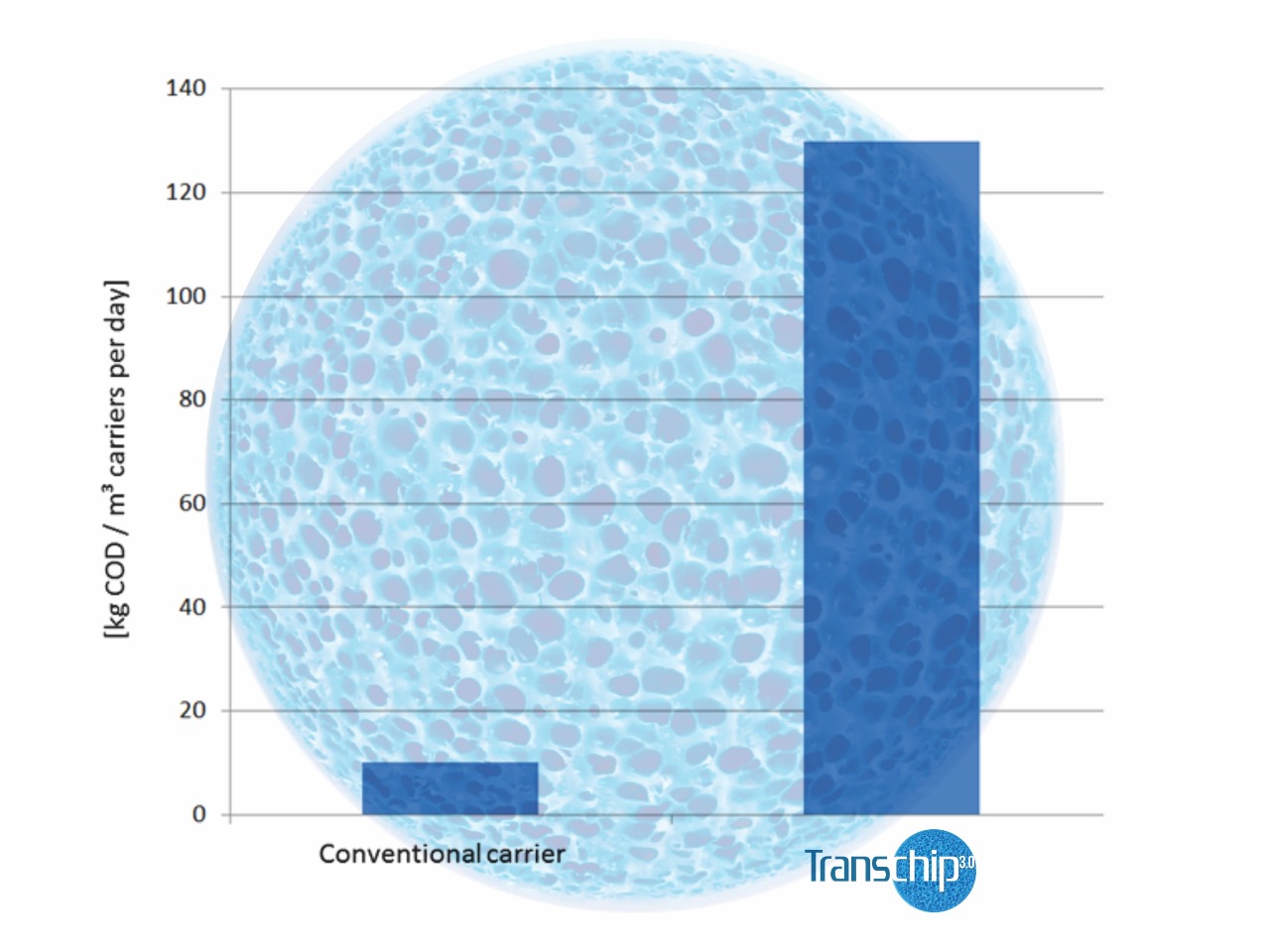
- Based on the positive findings gained here, the Transchip 3.0 volume was increased in a second step to 11% related to the “conventional”carrier volume that would normally have been required. Figure no. 8 shows the results that were obtained afterwards. It is clearly obvious that the Transchip 3.0 reactor now attained the same removal efficiency as the old reactor did, so there was no need to further increase the Transchip 3.0 volume. In direct comparison with the conventional carrier media, the increase in the degradation efficiency by the factor of nearly 10 achieved by Transchip 3.0 was proven clearly and impressively. Based on both these findings and the stable operating results of the new Transchip 3.0 high-load stage, the old reactor was put out of operation and modified.
- Now, both stages are operated with the Transchip 3.0 technology and reliably attain the required treated effluent parameters. With regard to the scope of the entire upgrading, it is worth mentioning for the sake of completeness that at least 1,000 m³/h of sewage water will be treated herein the future whereas an upgrade of the low-load activated sludge stage is also envisaged. As a second example and as a further proof of the efficiency of the Transchip 3.0 also in nitrogen elimination, its use in the nitrification stage of a coke oven effluent treatment plant will be described in the following. Coke oven effluents are not only regarded as difficult to treat –acc. to our experience they can be biologically purifiedby means of multiple biological stage systems only.
- Here, particular biodegradation rate requirements have to be fulfilled; this applies especially when taking into consideration the reactor sizes that need to remain within implementable as well as controllable dimensions. Such like plants using Transchip 3.0 carriers have been in continuous operation for more than four years. Converted into population equivalents, the largest of them have a nitrification capacity of around 100,000 PE and 55,000 PE. The Transchip 3.0’s superiority can be observed here, too. Removal rates of 4-5 kg NH4-N per m³ of carrier volume are reliably attained at a constant level, even though the Transchip 3.0 reactors are smaller by a factor of 5 than the activated sludge tanks which are usually required.